Batteries are the most expensive consumable component in an off-grid solar system. While solar panels can last 25+ years with minimal degradation, batteries are chemical devices that wear out over time.
Understanding why they degrade and how fast they degrade is crucial for protecting your investment.
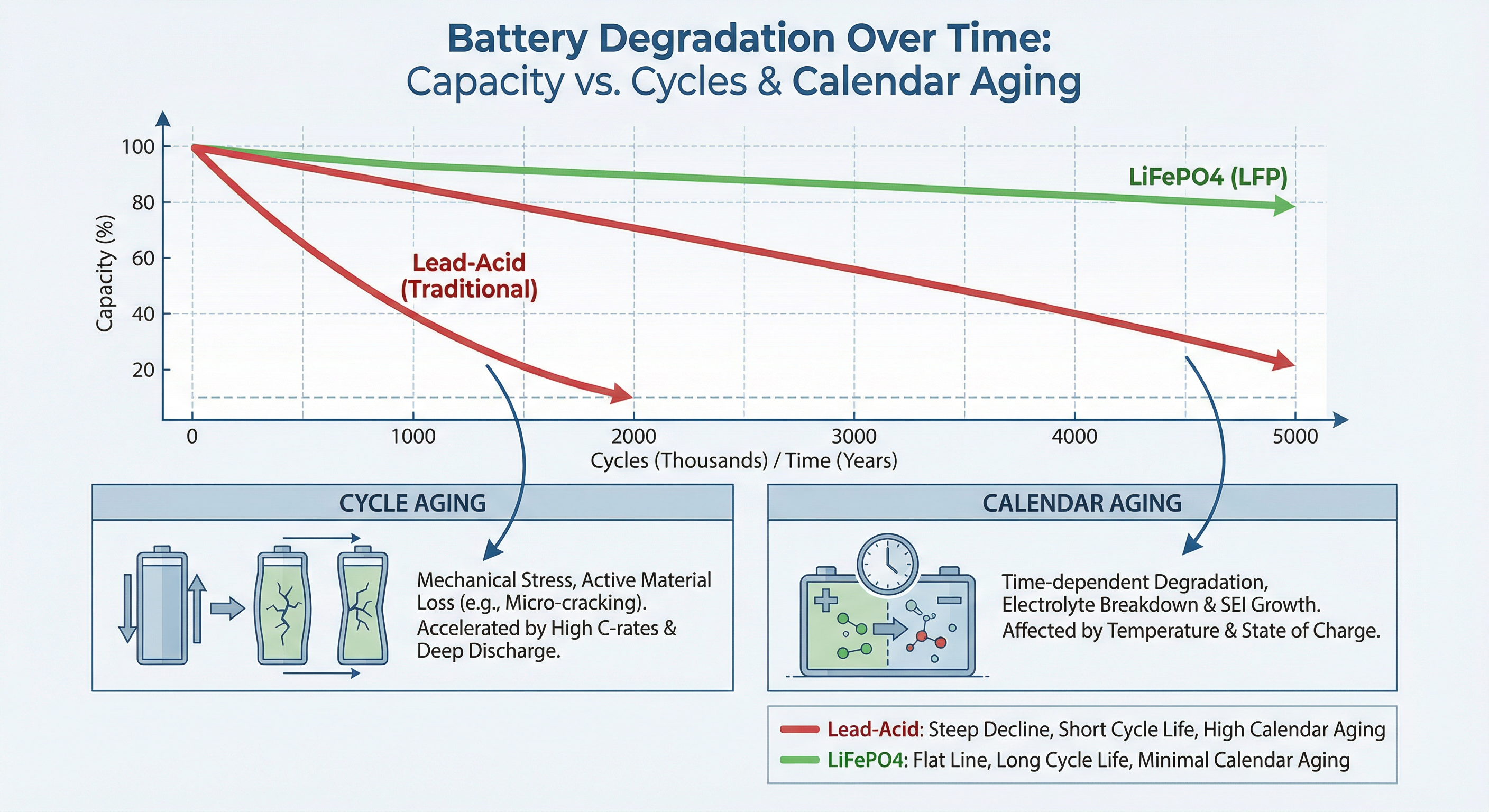
In 2026, the two main chemistries—Lead Acid and Lithium Iron Phosphate (LiFePO4)—behave very differently.
1. Cycle Life: The "Gas Tank" Analogy
Every time you discharge a battery and recharge it, that is one "cycle." Batteries are rated for a certain number of cycles before they lose significant capacity (usually down to 80% of original capacity).
Lead Acid (AGM/Gel)
- Typical Cycle Life: 300 - 500 cycles at 50% Depth of Discharge (DoD).
- Real World: If you cycle them daily to 50%, they will last 1 - 2 years. If you only discharge them 20%, they might last 5 years.
- The "Death Spiral": As they age, their capacity shrinks. A 100Ah battery becomes a 90Ah battery, then 80Ah. This means you discharge them deeper each night to get the same energy, accelerating the wear.
Lithium (LiFePO4)
- Typical Cycle Life: 3000 - 6000+ cycles at 80% DoD.
- Real World: If you cycle them daily to 80%, they will last 10 - 15 years.
- Degradation Curve: They degrade very slowly and linearly. You won't notice a drop in capacity for years.
2. Depth of Discharge (DoD)
This is the single biggest factor you can control.
- Lead Acid: Never go below 50%. Going to 80% DoD (20% remaining) can kill a lead-acid battery in less than 100 cycles.
- Lithium: Can go to 80-90% routinely. Going to 100% (0% remaining) occasionally is fine, but constantly sitting at 0% or 100% can stress the chemistry slightly.
Pro Tip: Oversizing your battery bank makes it last longer. If you need 5kWh, buying a 10kWh bank means you only discharge it 50%, doubling (or tripling) its lifespan.
3. Temperature: The Silent Killer
Batteries are like Goldilocks; they like it "just right" (roughly 25°C / 77°F).
Heat
- Lead Acid: Every 8°C (15°F) increase above 25°C cuts battery life in half. A battery kept at 95°F will last half as long as one at 77°F.
- Lithium: High heat degrades them too, but they are more resilient than lead acid. However, prolonged exposure to >45°C (113°F) is bad.
Cold
- Lead Acid: Capacity drops temporarily (sluggish chemical reaction). At freezing, a 100Ah battery might only act like a 70Ah battery. But it doesn't permanently damage it unless it freezes (which happens if discharged).
- Lithium: DO NOT CHARGE BELOW FREEZING. Charging LiFePO4 below 0°C causes lithium plating, which permanently damages the cell and can cause shorts. Discharging is fine down to -20°C.
Solution: Insulate your battery box. Use heating pads for lithium in winter.
4. C-Rate (Charging/Discharging Speed)
Charging or discharging too fast generates heat and stress.
- Lead Acid: Likes slow, steady currents (C/10 or C/20). Rapid charging boils the electrolyte.
- Lithium: Can handle high currents (1C), but prefers slower (0.5C or less).
Conclusion
To maximize battery life:
- Keep them cool (but not freezing).
- Oversize the bank to reduce Depth of Discharge.
- Switch to LiFePO4 if you can afford the upfront cost; the cost-per-cycle is vastly lower.
For a direct comparison of technologies, see Best Batteries for Off-Grid Solar: LiFePO4 vs Lead Acid.


